Silq's ClearTract®: Foley Catheter Innovation Unleashed
ClearTract® by Silq Technologies is an indwelling foley catheter that uses a zwitterionic surface to reduce infections and provide a more comfortable experience.
After spinal cord injury, using a urinary catheter to empty your bladder often becomes a part of daily life. A catheter is a hollow, flexible tube that is inserted into the bladder, either through the urethra or through the abdomen (suprapubic placement).
Catheter users choose between two types of catheters: intermittent or indwelling (also known as Foley). Intermittent catheters are used four to six times a day and disposed of after each use. Foley catheters remain in the body for longer periods and are typically exchanged every month.
SCI patients who have difficulty inserting catheters multiple times a day, or who are inconvenienced by the frequency, often choose a Foley catheter. However, due to their longer stay in the body, patients who use Foley catheters are at higher risk of infection and other catheter-related complications.

The most common Foley catheters are made from latex or silicone. These rubbers attract biological material to the catheter surface and initiate the formation of biofilm within minutes. Biofilms are the primary cause of reoccurring infection and are also directly linked to encrustation and blockage of catheters. The tacky, rubber catheter surface can also cause trauma to the bladder and urinary tract during use.
Current Foley catheter options to treat catheter-related complications
To address complications due to biofilm growth and infection, catheter manufacturers have incorporated antibiotics or silver technology into Foley catheters. Several studies have demonstrated that impregnation of antibiotics or silver may reduce bacteria in the short-term but have not noted any clinical benefit or improvement over time in reducing catheter-related complications.
Evidence of success of the preventative use of antibiotics to treat catheter-associated complications is inconclusive, and the overuse of broad-spectrum antibiotics have created new strains of multi-drug resistant microbes. This has caused serious concern and as a result, the Society for Healthcare Epidemiology of America advises against the routine use of antimicrobial impregnated catheters.
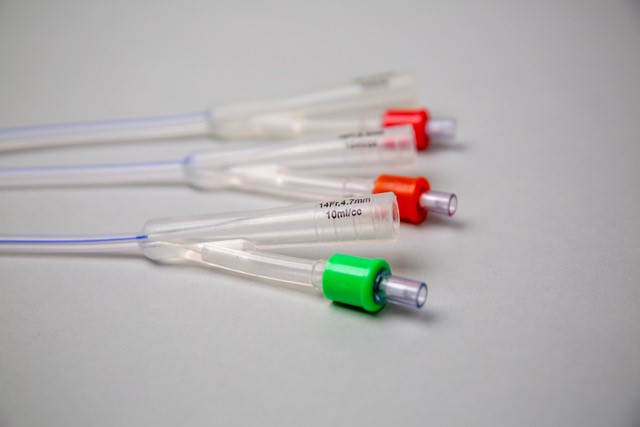
A new Foley catheter option using novel technology
ClearTract® is a new catheter recently introduced by Silq Technologies to address the issue of infection and catheter-related complications. Instead of using antibiotics to kill infectious microbes, ClearTract® catheters have a special a zwitterionic surface coating that is known to resist their deposition and growth.
Emulating the same biochemistry that surrounds every cell in our body, the surface treatment creates an extremely thin hydration layer that resists biofilm formation and confers greater lubricity, making implant and explant of the catheter easier and more comfortable. ClearTract® catheters have recently gained FDA clearance for urethral, suprapubic and nephrostomy placement. Patient and physician experience with the device has been overwhelmingly positive, with several examples of life-changing treatment for patients and their families.
If you are interested in this new catheter option, please consult with your physician to see if ClearTract® is right for you. Additional information, including patient and physician experiences with ClearTract® is available at Silq Technologies’ website: http://www.silq.tech. Patients interested in trying ClearTract® can also find the catheters available online at healthproductsforyou.com or shopcatheters.com (prescription required).
Stay Updated on Advancements On Traumatic Brain &
Spinal Cord Injuries
About the Author